The 3 Q's in Computer System Validation - IQ OQ PQ - eLeaP
4.8 (363) · € 26.00 · En Stock
Computer System Validation made easy with IQ-OQ-PQ. Validation in FDA-regulated orgs is crucial. Installation, Operational, Performance.

IQ, OQ, PQ - A Validation Process in the Medtech Industry - Elos Medtech
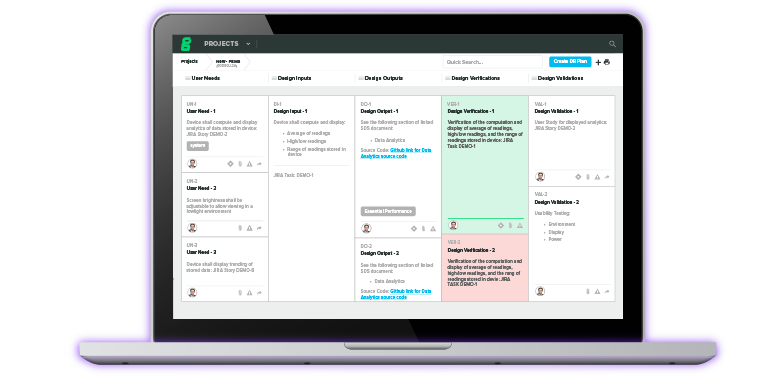
IQ, OQ, PQ: A Quick Guide to Process Validation
Fiverr freelancer will provide Technical Writing services and develop validation documentations vmp, urs, frs, ds, iq, oq, pq, sop, sr, tm including

develop validation documentations vmp, urs, frs, ds, iq, oq, pq, sop, sr, tm
Comply with 21 CFR Part 11: What it Means and How to Achieve It - eLeaP

Ensuring Data Integrity: A Comprehensive Guide to GxP Compliance in Regulated Industries
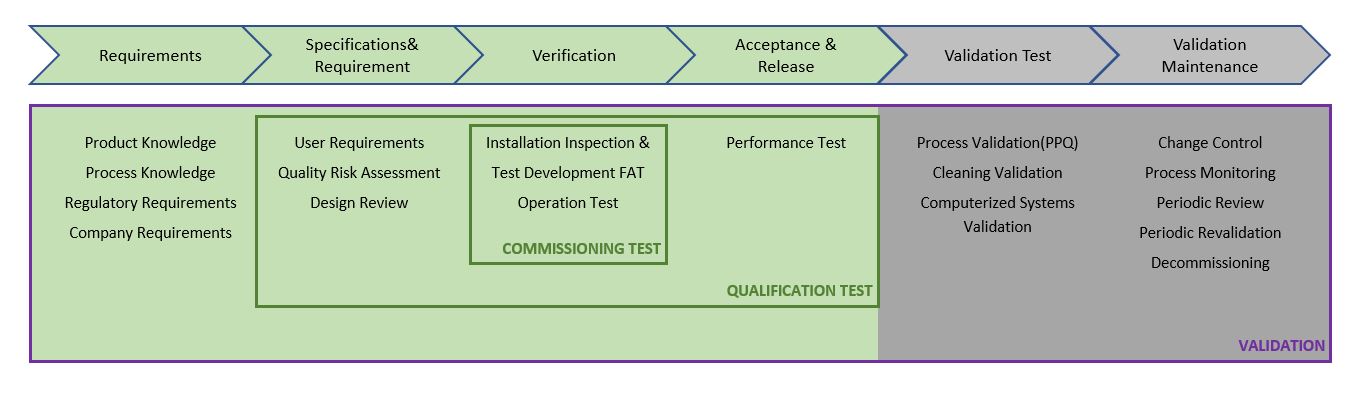
The Q's In Computer System Validation IQ OQ PQ ELeaP, 59% OFF
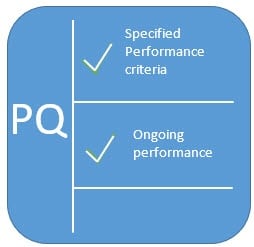
What Are IQ OQ PQ, The 3 Q's Of Software Validation Process

Life Sciences - eLeaP

What is IQ OQ PQ in Software Validation?












